|
These elements have some of the classic characteristics of the transition metals, but they tend to be softer and conduct more poorly than other transition metals. The post-transition metals are aluminum (Al), gallium (Ga), indium (In), thallium (Tl), tin (Sn), lead (Pb) and bismuth (Bi), and they span Group 13 to Group 17. 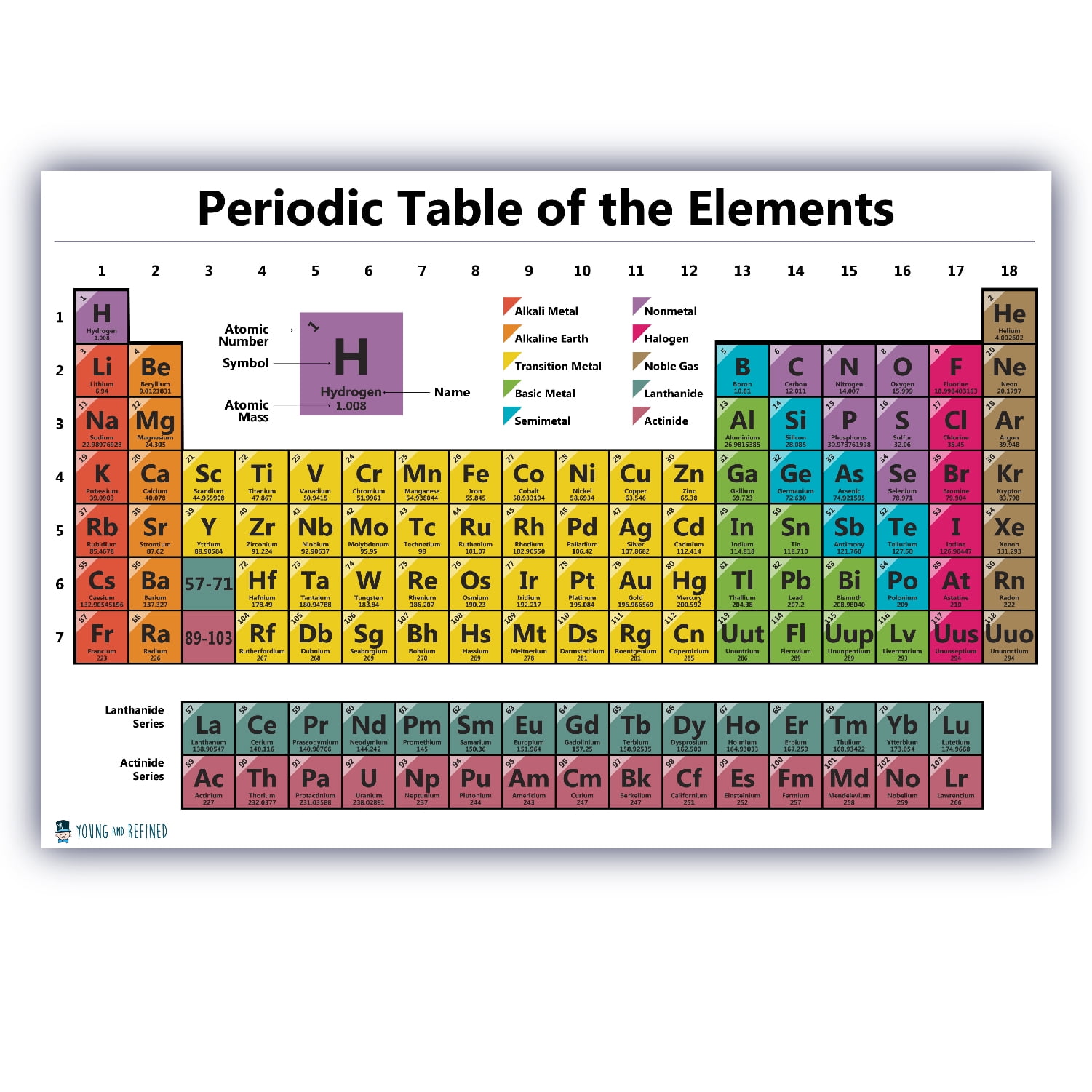
Post-transition metals: Ahead of the jump into the nonmetal world, shared characteristics aren't neatly divided along vertical group lines. 
Many of the greatest hits of the metal world - including gold, silver, iron and platinum - live here. Hard but malleable, shiny, and possessing good conductivity, these elements are what you typically think of when you hear the word metal. Transition metals: Returning to the main body of the table, the remainder of Groups 3 through 12 represent the rest of the transition metals. The actinides and the lanthanides together form a group called the inner transition metals. Of these elements, only thorium (Th) and uranium (U) occur naturally on Earth in substantial amounts. The elements in this group have a silvery white color and tarnish on contact with air.Īctinides: The actinides line the bottom row of the island and comprise elements 89, actinium (Ac), through 103, lawrencium (Lr). This is the lanthanides, elements 57 through 71 - lanthanum (La) to lutetium (Lu). Lanthanides: The third group is much too long to fit into the third column, so it is broken out and flipped sideways to become the top row of the island that floats at the bottom of the table. Their chemical reactions typically occur more slowly and produce less heat compared to the alkali metals. But they're not as reactive as the alkali metals. Each of these elements has two electrons in its outermost energy level, which makes the alkaline earths reactive enough that they're rarely found alone in nature. Hydrogen, with its single electron, also lives in Group 1, but the gas is considered a nonmetal.Īlkaline-earth metals: The alkaline-earth metals make up Group 2 of the periodic table, from beryllium (Be) through radium (Ra).  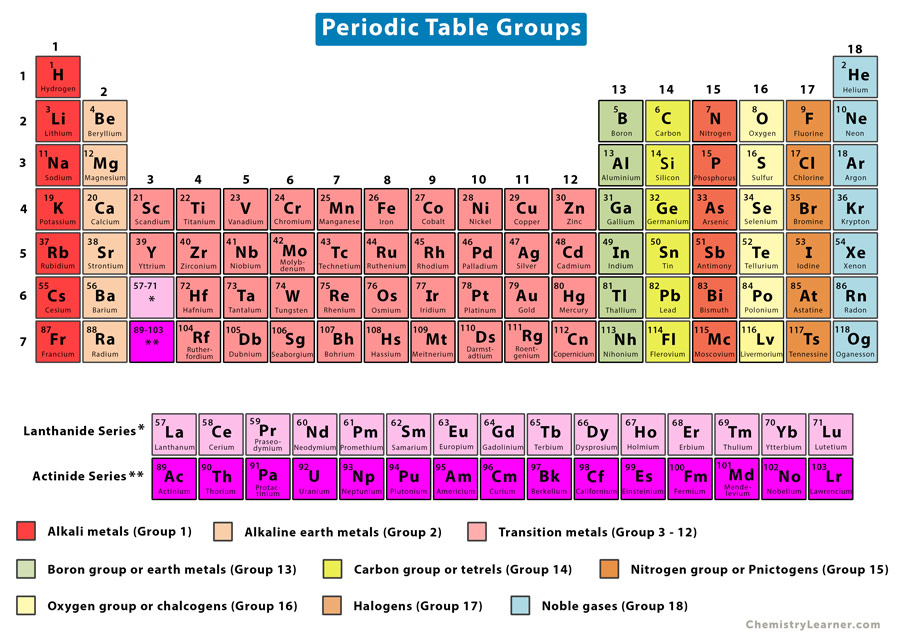
They are also extremely reactive and will burst into flame or even explode on contact with water, so chemists store them in oils or inert gases. Shiny and soft enough to cut with a knife, these metals start with lithium (Li) and end with francium (Fr). What do these electrons combine with in order to produce an energy-carrying molecule? Are paper towels combustible? What is the effect of pressure in the solubility of gas in liquid? How fast does baking soda dissolve in water? Why is sulfuric acid used on dead bodies such as Czar Nicholas II and his family? Which has more valence electrons atom or aluminum or phosphorus? What is another name for metalloid? How many moles of water molecules h2o are present in a 27 rams sample of water? Calculate the volume occupied by 2.88 moles of hydrogen at stp? How many milligram is a kilogram? What homogeneous example? what is the most reactive (non-metals) group in the periodic table? What is element management? What is the meaning of bis in gold purity? How many elements are in NH4? What charge does each subatomic particle carry? What is the ionic bond of S and Br? Why small quantity of chlorine fills the whole vessel in which it is enclosed? What is a solution that has a pH of 6 called? How many milligrams equals 2.The periodic table of elements is arranged into several broad groups (Image credit: Future) Groups of the Periodic tableĪlkali metals: The alkali metals make up most of Group 1, the table's first column. Trending Questions Electrons are excited in photosystem I.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
